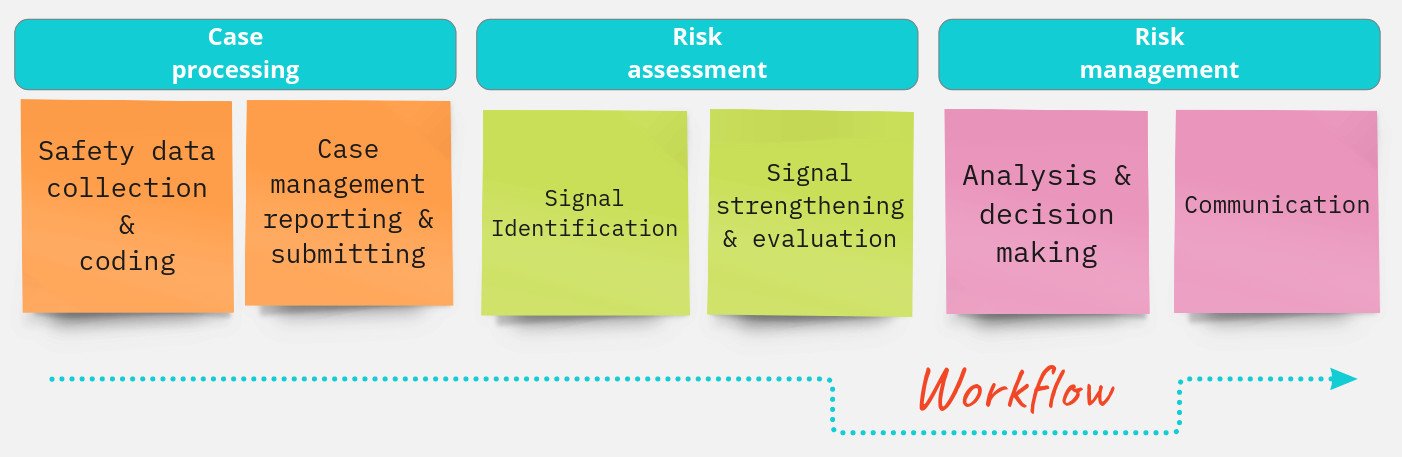
Even if you are still in the planning stages of your project, you are more than welcome to submit a request to us. We can assist you with a quotation or create a proposal for you based on the minimum of information you can provide.
Follow us
CEBIS Interantional
Romania- Helios Business Center (HBC),
47 Bd. Theodor Pallady,
Bucharest, Romania